 
Potassium is the most reactive, immediately produces purple sparks and flames.īe sure to use small pea-sized pieces of metal. Be careful of larger chunks, which, due to internal air pockets, may take off like rockets from dish. Optional: Use hexane to rinse mineral oil from metal surface. The metal will scoot around on the surface of the water, producing bubbles of hydrogen and causing the phenolphthalein indicator to turn pink. Three crystallizing dishes filled with waterĭrop a small piece of Li, Na or K into each dish.There are often microscopic pits in the metal causing the metal to launch. 
Do not do this experiment on the overhead. What is reactivity series and which is the most reactive metal Rubidium (Rb).The metals react vigorously with water such that even water in the air is enough to ignite sodium and potassium.Store all metals under oil to protect from moisture.Increasing reactivity of group one metals is demonstrated. The resulting metal hydroxide solutions change the indicator from clear to pink. Lithium, sodium and potassium metals are added to water containing phenolphthalein indicator. Facility Announcements & Equipment Logbooks.Additional Info for Prospective Students.It has to be kept in dry mineral oil or in a hydrogen atmosphere. It is named after the red colour that can been seen in a spectrometer. It was discovered in 1861 by Robert Bunsen and Gustav Kirchhoff. When it reacts with water, it releases hydrogen gas, which then catches fire. Rubidium reacts spontaneously in air and water. Its other main use is as an alloying metal to make platinum harder.Įlement 37 on the periodic table. Be careful of larger chunks, which, due to internal air. If No single replacement reaction occurs, write NR to the right of the arrow. Optional: Use hexane to rinse mineral oil from metal surface. Using the Activity Series Table, complete the following reactions by writing the products that are formed. It is often electroplated as a coating onto other metals to make a shiny, non-tarnishing finish on tableware and jewellery. Drop a small piece of Li, Na or K into each dish. A copper strip placed in a silver nitrate solution causes the silver to be precipitated onto the copper strip. Reactivity series – Copper is more reactive than silver. This important reaction is used in refining some metals. Reactivity series – Because metals differ in their reactivity, if a more reactive metal is placed in a solution of a less reactive metal compound, a reaction occurs that takes the reactive metal into solution and forces the less reactive metal out of solution, forming a precipitate. Rhodium is normally obtained as a by-product from the refining of copper and nickel. What is the reactivity of rubidium how is the reactivity for ptassium. A rare, silver-white member of the transition metals. the reactivity series is a type of chemical 'league table' using critiria that sciance agree about. It has few direct uses.Įlement 45 on the periodic table. Rank the following elements from the most reactive to least reactive. Specifically, use it for identifying whether a metal reacts with water or acid or whether it replaces another metal in a reaction. Knowing the activity series helps you predict whether or not a chemical reaction occurs. It was discovered in 1925 by the German chemists Ida and Walter Noddack and Otto Carl Berg. The activity series of metals or reactivity series is a list of metals from most reactive to least reactive. 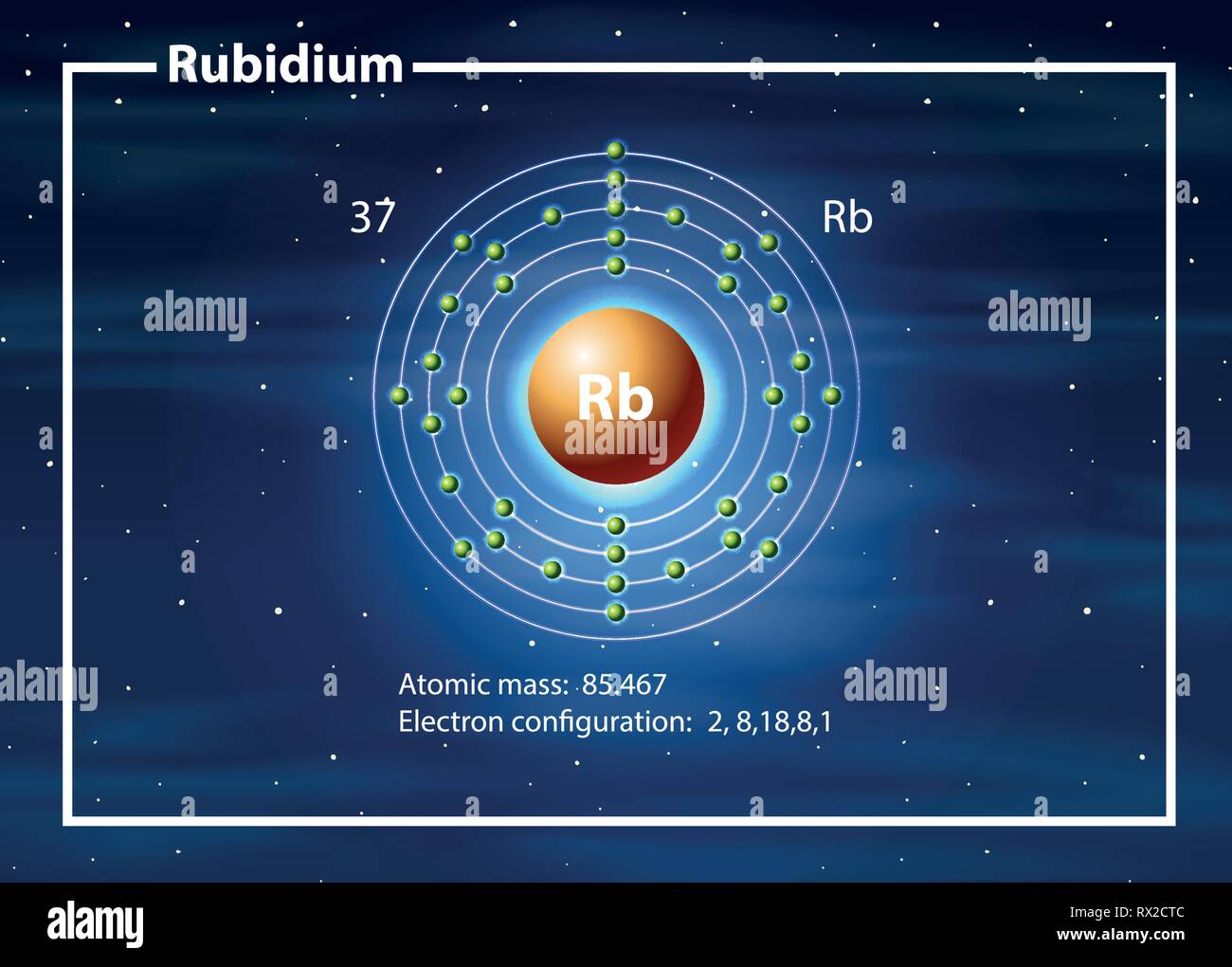
A very dense element and one of the transition metals. For example, the approximate relative atomic mass of chlorine is 35, calcium 40 and gold 197.Įlement 75 on the periodic table. If the relative atomic mass is given as a rounded figure, it is called an approximate relative atomic mass. Nowadays, a measure of the mass of an atom is made relative to the mass of one-twelfth of an atom of carbon-12. The study programs for scientific and technical degrees include classroom theoretical teaching, analysis and resolution of real exercises, and some more practical teaching in the laboratory. In the past, a measure of the mass of an atom on a scale relative to the mass of an atom of hydrogen, where hydrogen has an atomic mass of 1. To remove oxygen from or add hydrogen to a compound. Predict which method would be used to extract cobalt from its ores. Hydrogen is usually included in the series for comparative purposes. The metal cobalt is between zinc and lead in the Reactivity Series. In order of their reactivity, with the most reactive metals, such as potassium, at the top and the least reactive metals, such as platinum, at the bottom. Whats the reactivity series from most reactive to least reactive. Metals are arranged in the reactivity series. Lead Hydrogen Copper Mercury Silver Gold Platinum Potassium Sodium Calcium Magnesium Aluminium Manganese Chromium Zinc 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
